In a groundbreaking study by AlphaFold, scientists mapped the structure of a large protein that gives us the shape of “bad cholesterol.” This discovery could help transform the way researchers and clinicians treat the world’s leading cause of death.
The race to uncover the key proteins behind heart disease has long been both an important public health goal and a stubborn scientific problem.
For University of Missouri (Mizzou) assistant professors Zachary Berntsen and Keith Cassidy, it was also personal. Both have a family history of heart disease. It’s a reminder of what’s at stake in their work to better understand and ultimately help cure this deadly disease.
“For 50 years, people have wanted to know what this protein looks like,” Bernsen says.
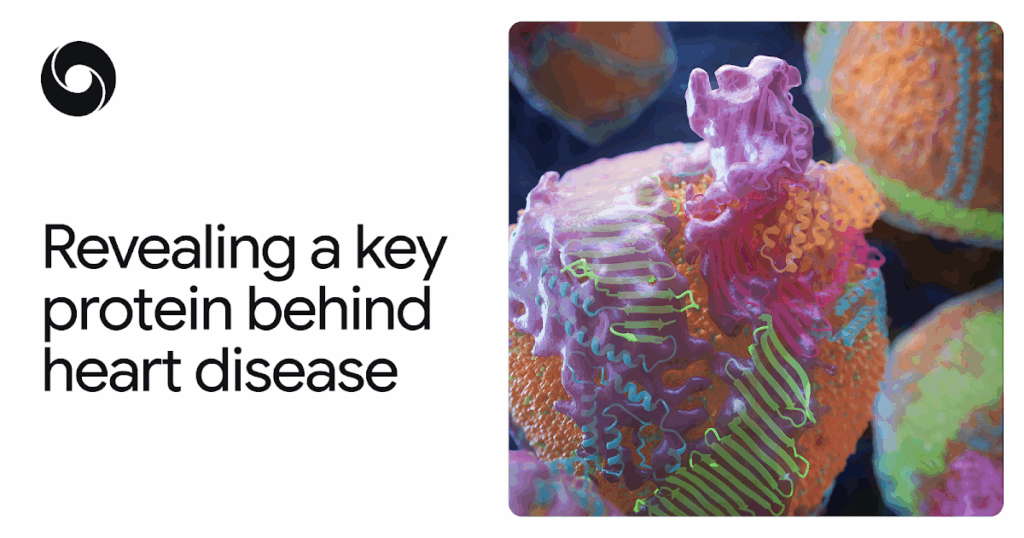
That protein, apoB100, has defied mapping not only because it is large (for a protein) but also because it binds to fats and other molecules in complex ways. ApoB100 forms the molecular scaffold of “bad cholesterol”, known to scientists as low-density lipoprotein (LDL).
LDL is the major carrier of fat through the bloodstream and is an important risk factor for atherosclerotic cardiovascular disease (ASCVD), the leading cause of death worldwide. It is hoped that discovering the structure of this key protein will shed light on how bad cholesterol can be harmful in the body, giving scientists a better opportunity to develop ways to prevent and treat ASCVD. AlphaFold is playing a central role in this effort.
At Mizzou, biochemist Berndsen first used cryo-electron microscopy (cryo-EM) to capture images of LDL particles. The images weren’t sharp enough to map apoB100’s structure with atomic precision, so Cassidy, Bernsen’s physicist collaborator, turned to AlphaFold. He used it to predict protein structures at atomic resolution and refined the predicted shapes by comparing them to cryo-EM image data.
Cassidy says that tackling the problem using both a cryo-EM microscope and Alphafold led to this breakthrough. “AlphaFold played a key role in this discovery, providing the raw material to interpret experimental structures in ways that frankly were not possible before.”
The resulting model revealed the key proteins of bad cholesterol in surprising detail. That is, a cage-like shell that wraps around each LDL particle, including a ribbon-like belt that keeps the particle intact in the bloodstream. Knowledge of this structure opens new possibilities for the prevention, diagnosis, and treatment of high cholesterol and ASCVD, including therapies that more precisely target LDL. The potential benefits to global health cannot be overstated.
Although such applications take time, elucidating the structure of apoB100 is a breakthrough and extremely gratifying for Berntsen. “This was the first structure I looked at the AlphaFold the week it became available, and the first protein I wanted to look at in a two-story cryo-EM machine,” he says. “Elucidating the structure of apoB100 was a dream come true.”